104b-pf00092-mtr-pfem-en-002
Dieses Dokument ist Teil der Anfrage „Briefverkehr mit BioNtech“
SAFETY DATA SHEET
Revision date 07-Dec-2021 Version 3 Page 1 / 12
Section 1: IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE
COMPANY/UNDERTAKING
1.1. Product identifier
Product Name Pfizer-BioNTech COVID-19 Vaccine
Product Code(s) PF00092
Form nanoform
Synonyms Comirnaty; PF-07302048 containing PF-07305885 (BNT162b2); CorVAC Containing
PF-07305885 (BNT162b2) ; CoVVAC Containing PF-07305885 (BNT162b2); COVID
Vaccine Containing PF-07305885 (BNT162b2); COVID-19 Vaccine Containing
PF-07305885 (BNT162b2)
Trade Name: Not applicable
Compound Number PF-07302048
Item Code H000022941: H000023057;H000024547: H000024742
Chemical Family: Lipid Nanoparticles containing PF-07305885 (BNT162b2) and Lipids
1.2. Relevant identified uses of the substance or mixture and uses advised against
Recommended Use Pharmaceutical product
1.3. Details of the supplier of the safety data sheet
Pfizer Inc Pfizer Ireland Pharmaceuticals
235 East 42nd Street OSG Building
New York, New York 10017 Ringaskiddy, Co. Cork.
1-800-879-3477 Ireland
+353 21 4378701
1.4. Emergency telephone number
Emergency Telephone Chemtrec 1-800-424-9300 International Chemtrec (24 hours):+1-703-527-3887
E-mail address pfizer-MSDS@pfizer.com
Section 2: HAZARDS IDENTIFICATION
2.1. Classification of the substance or mixture
GHS - Classification: Not classified as hazardous
2.2. Label elements
Signal word Not classified
Hazard statements Not classified in accordance with international standards for workplace safety.
2.3. Other hazards
Other hazards An Occupational Exposure Value has been established for one or more of the ingredients
(see Section 8).
Note: This document has been prepared in accordance with standards for workplace safety,
which require the inclusion of all known hazards of the product or its ingredients regardless
_____________________________________________________________________________________________
PF00092

SAFETY DATA SHEET
Product Name Pfizer-BioNTech COVID-19 Vaccine Page 2 / 12
Revision date 07-Dec-2021 Version 3
_____________________________________________________________________________________________
of the potential risk. The precautionary statements and warnings included may not apply in
all cases. Your needs may vary depending upon the potential for exposure in your
workplace.
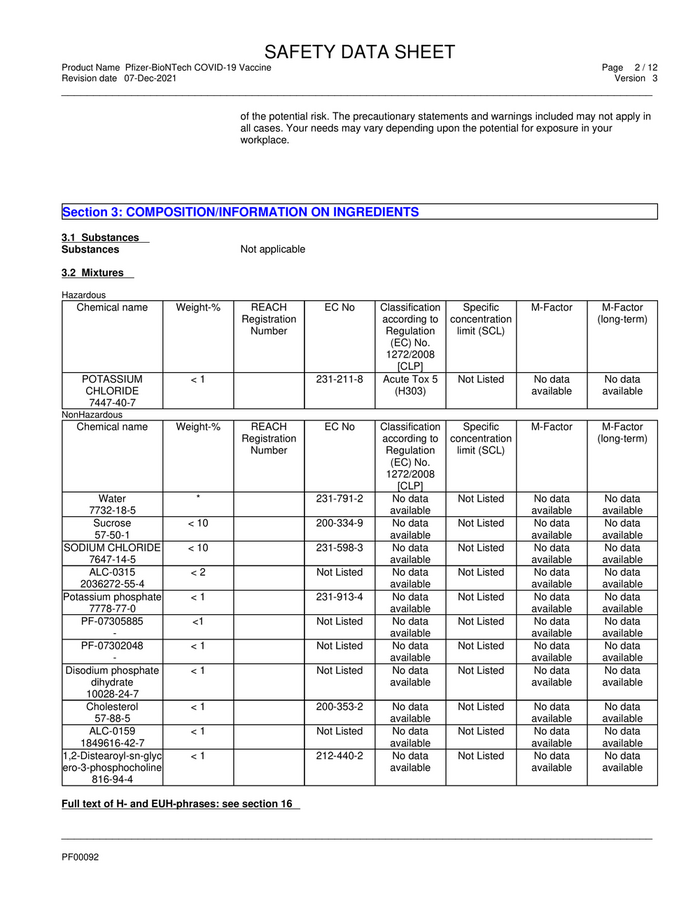
Section 3: COMPOSITION/INFORMATION ON INGREDIENTS
3.1 Substances
Substances Not applicable
3.2 Mixtures
Hazardous
Chemical name Weight-% REACH EC No Classification Specific M-Factor M-Factor
Registration according to concentration (long-term)
Number Regulation limit (SCL)
(EC) No.
1272/2008
[CLP]
POTASSIUM <1 231-211-8 Acute Tox 5 Not Listed No data No data
CHLORIDE (H303) available available
7447-40-7
NonHazardous
Chemical name Weight-% REACH EC No Classification Specific M-Factor M-Factor
Registration according to concentration (long-term)
Number Regulation limit (SCL)
(EC) No.
1272/2008
[CLP]
Water * 231-791-2 No data Not Listed No data No data
7732-18-5 available available available
Sucrose < 10 200-334-9 No data Not Listed No data No data
57-50-1 available available available
SODIUM CHLORIDE < 10 231-598-3 No data Not Listed No data No data
7647-14-5 available available available
ALC-0315 <2 Not Listed No data Not Listed No data No data
2036272-55-4 available available available
Potassium phosphate <1 231-913-4 No data Not Listed No data No data
7778-77-0 available available available
PF-07305885 <1 Not Listed No data Not Listed No data No data
- available available available
PF-07302048 <1 Not Listed No data Not Listed No data No data
- available available available
Disodium phosphate <1 Not Listed No data Not Listed No data No data
dihydrate available available available
10028-24-7
Cholesterol <1 200-353-2 No data Not Listed No data No data
57-88-5 available available available
ALC-0159 <1 Not Listed No data Not Listed No data No data
1849616-42-7 available available available
1,2-Distearoyl-sn-glyc <1 212-440-2 No data Not Listed No data No data
ero-3-phosphocholine available available available
816-94-4
Full text of H- and EUH-phrases: see section 16
_____________________________________________________________________________________________
PF00092
SAFETY DATA SHEET
Product Name Pfizer-BioNTech COVID-19 Vaccine Page 3 / 12
Revision date 07-Dec-2021 Version 3
_____________________________________________________________________________________________
Acute Toxicity Estimate
Chemical name Oral LD50 Dermal LD50 Inhalation LC50 - 4 Inhalation LC50 - 4 Inhalation LC50 - 4
hour - dust/mist - hour - vapor - mg/L hour - gas - ppm
mg/L
Water 89838.9 No data available No data available No data available No data available
7732-18-5
Sucrose 29700 No data available No data available No data available No data available
57-50-1
SODIUM CHLORIDE 3000 10000 No data available No data available No data available
7647-14-5
Potassium phosphate 3200 No data available 0.83 No data available No data available
7778-77-0
POTASSIUM CHLORIDE 3020 No data available No data available No data available No data available
7447-40-7
Cholesterol No data available 2000 No data available No data available No data available
57-88-5
Additional information - Not Assigned
* Proprietary
Non-hazardous ingredients provided for completeness. Ingredient(s) indicated as
hazardous have been assessed under standards for workplace safety. In accordance with
29 CFR 1910.1200, the exact percentage composition of this mixture has been withheld as
a trade secret.
Section 4: FIRST AID MEASURES
4.1. Description of first aid measures
Inhalation Remove to fresh air. Seek immediate medical attention/advice.
Eye contact Rinse thoroughly with plenty of water for at least 15 minutes, lifting lower and upper eyelids.
Consult a physician.
Skin contact Remove contaminated clothing. Flush area with large amounts of water. Use soap. Seek
medical attention.
Ingestion Never give anything by mouth to an unconscious person. Wash out mouth with water. Do
not induce vomiting unless directed by medical personnel. Seek medical attention
immediately.
4.2. Most important symptoms and effects, both acute and delayed
Most important symptoms and For information on potential signs and symptoms of exposure, See Section 2 - Hazards
effects Identification and/or Section 11 - Toxicological Information.
4.3. Indication of any immediate medical attention and special treatment needed
Note to physicians None.
Section 5: FIRE-FIGHTING MEASURES
5.1. Extinguishing media
Suitable Extinguishing Media Dry chemical, CO2, alcohol-resistant foam or water spray.
_____________________________________________________________________________________________
PF00092

SAFETY DATA SHEET
Product Name Pfizer-BioNTech COVID-19 Vaccine Page 4 / 12
Revision date 07-Dec-2021 Version 3
_____________________________________________________________________________________________
5.2. Special hazards arising from the substance or mixture
Specific hazards arising from the Fine particles (such as mists) may fuel fires/explosions.
chemical
Hazardous combustion products Formation of toxic gases is possible during heating or fire.
5.3. Advice for firefighters
Special protective equipment for Firefighters should wear self-contained breathing apparatus and full firefighting turnout
fire-fighters gear. Use personal protection equipment.
Section 6: ACCIDENTAL RELEASE MEASURES
6.1. Personal precautions, protective equipment and emergency procedures
Personal precautions Personnel involved in clean-up should wear appropriate personal protective equipment (see
Section 8). Minimize exposure.
For emergency responders Use personal protection recommended in Section 8.
6.2. Environmental precautions
Environmental precautions Place waste in an appropriately labeled, sealed container for disposal. Care should be
taken to avoid environmental release.
6.3. Methods and material for containment and cleaning up
Methods for containment Prevent further leakage or spillage if safe to do so.
Methods for cleaning up Contain the source of spill if it is safe to do so. Collect spill with absorbent material. Clean
spill area thoroughly.
Prevention of secondary hazards Clean contaminated objects and areas thoroughly observing environmental regulations.
6.4. Reference to other sections
Reference to other sections See section 8 for more information. See section 13 for more information.
Section 7: HANDLING AND STORAGE
7.1. Precautions for safe handling
Advice on safe handling
Restrict access to work area. A change area to facilitate 'good laboratory/manufacturing' decontamination practices is
recommended. Additional controls (based on risk assessment) should be implemented where open handling is required. Use
enclosed manufacturing processing strategies. Avoid inhalation and contact with skin, eye, and clothing. When handling, use
appropriate personal protective equipment (see Section 8). Wash hands and any exposed skin after removal of PPE. Releases to
the environment should be avoided. Review and implement appropriate technical and procedural waste water and waste disposal
measures to prevent occupational exposure or environmental releases.
General hygiene considerations Handle in accordance with good industrial hygiene and safety practice.
7.2. Conditions for safe storage, including any incompatibilities
Storage Conditions Store as directed by product packaging.
7.3. Specific end use(s)
Specific use(s) Vaccine.
Section 8: EXPOSURE CONTROLS/PERSONAL PROTECTION
_____________________________________________________________________________________________
PF00092

SAFETY DATA SHEET
Product Name Pfizer-BioNTech COVID-19 Vaccine Page 5 / 12
Revision date 07-Dec-2021 Version 3
_____________________________________________________________________________________________
8.1. Control parameters
Exposure Limits
Refer to available public information for specific member state Occupational Exposure Limits.
Sucrose
ACGIH TLV 10 mg/m3
Bulgaria 10.0 mg/m3
Estonia 10 mg/m3
France 10 mg/m3
Ireland 10 mg/m3
STEL: 20 mg/m3
Latvia 5 mg/m3
Spain 10 mg/m3
OSHA PEL 15 mg/m3
5 mg/m3
(vacated) TWA: 15 mg/m3 total dust
(vacated) TWA: 5 mg/m3 respirable fraction
United Kingdom TWA: 10 mg/m3
STEL: 20 mg/m3
SODIUM CHLORIDE
Latvia 5 mg/m3
Russia MAC: 5 mg/m3
Potassium phosphate
Russia MAC: 10 mg/m3
POTASSIUM CHLORIDE
Bulgaria 5.0 mg/m3
Latvia 5 mg/m3
Russia MAC: 5 mg/m3
Pfizer Occupational Exposure Band The Vaccines Occupational Exposure Band (V-OEB) is a classification that has been
(OEB) Statement: assigned to biotechnology-based vaccines and antigen components. Risk assessments
should be performed to assess potential exposures and determine appropriate controls.
The purpose of the Occupational Exposure Band (OEB) classification system is to separate
substances into different Hazard categories when the available data are sufficient to do so,
but inadequate to establish an Occupational Exposure Limit (OEL). The OEB given is based
upon an analysis of all currently available data; as such, this value may be subject to
revision when new information becomes available.
SODIUM CHLORIDE
Pfizer Occupational Exposure OEB 1 (control exposure to the range of 1000ug/m3 to 3000ug/m3)
Band (OEB):
ALC-0315
Pfizer Occupational Exposure OEB 3 - Contact Hazards Unknown (control exposure to the range of 10ug/m3 to <
Band (OEB): 100ug/m3)
POTASSIUM CHLORIDE
Pfizer Occupational Exposure OEB 1 (control exposure to the range of 1000ug/m3 to 3000ug/m3)
Band (OEB):
PF-07305885
Pfizer Occupational Exposure V-OEB
Band (OEB):
PF-07302048
Pfizer Occupational Exposure V-OEB
Band (OEB):
ALC-0159
Pfizer Occupational Exposure OEB 3 - Contact Hazards Unknown (control exposure to the range of 10ug/m3 to <
Band (OEB): 100ug/m3)
8.2. Exposure controls
Engineering controls Release prevention and exposure protection measures should be established for any
_____________________________________________________________________________________________
PF00092

SAFETY DATA SHEET
Product Name Pfizer-BioNTech COVID-19 Vaccine Page 6 / 12
Revision date 07-Dec-2021 Version 3
_____________________________________________________________________________________________
activities involving this material, as determined by a risk assessment conducted using
appropriate Occupational Hygiene Risk Assessment tools. The containment level required
for the activity should be based on the conclusions of the risk assessment. Where
warranted, engineering controls, such as biosafety cabinets, should be applied as the
primary means to control exposures.
Environmental exposure controls No information available.
Personal protective equipment Contact your safety and health professional or safety equipment supplier for assistance in
selecting the correct protective clothing/equipment based on an assessment of the
workplace conditions, other chemicals used or present in the workplace and specific
operational processes. Refer to applicable national standards and regulations in the
selection and use of personal protective equipment (PPE).
Eye/face protection Wear safety glasses as minimum protection (goggles recommended). (Eye protection
must meet the standards in accordance with EN166, ANSI Z87.1 or international
equivalent.).
Hand protection Wear impervious gloves, (e.g. Nitrile, etc.) to prevent skin contact. (Protective gloves must
meet the standards in accordance with EN374, ASTM F1001 or international equivalent.).
Skin and body protection Wear impervious disposable protective clothing when handling this compound. Full body
protection is recommended (scale dependent). (Protective clothing must meet the
standards in accordance with EN13982, ANSI 103 or international equivalent.).
Respiratory protection If operating and handling conditions result in airborne exposure, wear an appropriate
respirator with a protection factor sufficient to control exposures (e.g. particulate cartridge
with a full face respirator, P3 filter). (Respirators must meet the standards in accordance
with EN136, EN143, ASTM F2704-10 or international equivalent.).
General hygiene considerations Handle in accordance with good industrial hygiene and safety practice.
Section 9: PHYSICAL AND CHEMICAL PROPERTIES
9.1. Information on basic physical and chemical properties
Physical state Liquid
Color milky white
Odor No information available.
Odor threshold No information available
Molecular formula Mixture
Molecular weight Mixture
Property Values
pH 7.4
Melting point / freezing point No data available
Boiling point / boiling range
Flash point No information available
Evaporation rate No data available
Flammability (solid, gas) No data available
Flammability Limit in Air
Upper flammability limit: No data available
Lower flammability limit: No data available
Vapor pressure No data available
Vapor density No data available
Relative density No data available
Water solubility No data available
_____________________________________________________________________________________________
PF00092

SAFETY DATA SHEET
Product Name Pfizer-BioNTech COVID-19 Vaccine Page 7 / 12
Revision date 07-Dec-2021 Version 3
_____________________________________________________________________________________________
Solubility(ies) No data available
Partition coefficient No data available
Autoignition temperature No data available
Decomposition temperature No data available
Kinematic viscosity No data available
Dynamic viscosity No data available
Particle characteristics
Particle Size No information available
Particle Size Distribution No information available
Explosive properties No information available
9.2. Other information
No information available
9.2.1. Information with regard to physical hazard classes
No information available
9.2.2. Other safety characteristics
No information available
Section 10: STABILITY AND REACTIVITY
10.1. Reactivity
Reactivity No data available.
10.2. Chemical stability
Stability Stable under normal conditions.
Explosion data
Sensitivity to Mechanical Impact No data available.
Sensitivity to Static Discharge No data available.
10.3. Possibility of hazardous reactions
Possibility of hazardous reactions No information available.
10.4. Conditions to avoid
Conditions to avoid Fine particles (such as mists) may fuel fires/explosions. As a precautionary measure, keep
away from heat sources and electrostatic discharge.
10.5. Incompatible materials
Incompatible materials As a precautionary measure, keep away from strong oxidizers.
10.6. Hazardous decomposition products
Hazardous decomposition products No data available.
Section 11: TOXICOLOGICAL INFORMATION
11.1. Information on hazard classes as defined in Regulation (EC) No 1272/2008
General Information: Toxicological properties have not been thoroughly investigated. The following information is
available for the individual ingredients.
Short term In the event of accidental injection, an allergic reaction may occur. If an allergic reaction
occurs, the worker should be removed to the nearest emergency room and the appropriate
therapy instituted.
Known Clinical Effects: Based on clinical trials in humans, possible adverse effects following intravenous exposure
to this compound may include: injection site pain, muscle pain, headache, fever, chills,
tiredness, joint pain, abnormal redness of skin (erythema), and sleep disturbances. Serious
allergic reactions, including anaphylaxis, have been reported.
Acute Toxicity: (Species, Route, End Point, Dose)
_____________________________________________________________________________________________
PF00092

SAFETY DATA SHEET
Product Name Pfizer-BioNTech COVID-19 Vaccine Page 8 / 12
Revision date 07-Dec-2021 Version 3
_____________________________________________________________________________________________
Sucrose
Rat Oral LD 50 29,700 mg/kg
SODIUM CHLORIDE
Rat Sub-tenon injection (eye) LC50/1hr > 42 g/m3
Rat Oral LD 50 3 g/kg
Mouse Oral LD 50 4 g/kg
Rabbit Dermal LD 50 > 10 g/kg
POTASSIUM CHLORIDE
Rat Oral LD50 3020 mg/kg
Potassium phosphate
Rat Oral LD50 3200 mg/kg
Rabbit Dermal LC50 > 4640 mg/kg
Chemical name Oral LD50 Dermal LD50 Inhalation LC50
Water > 90 mL/kg ( Rat ) - -
Sucrose = 29700 mg/kg ( Rat ) - -
SODIUM CHLORIDE = 3 g/kg ( Rat ) > 10000 mg/kg ( Rabbit ) > 42 mg/L ( Rat ) 1 h
Potassium phosphate = 3200 mg/kg ( Rat ) - > 0.83 mg/L ( Rat ) 4 h
POTASSIUM CHLORIDE = 2600 mg/kg ( Rat ) - -
Cholesterol > 2000 mg/kg ( Rat ) -
Irritation / Sensitization: (Study Type, Species, Severity)
SODIUM CHLORIDE
Skin irritation Rabbit Mild
Eye irritation Rabbit Mild
POTASSIUM CHLORIDE
Eye Irritation Rabbit Mild
Repeated Dose Toxicity: (Duration, Species, Route, Dose, End Point, Target Organ)
PF-07302048
4 Week(s) Rat Intramuscular * 10 µg LOAEL Skin, Blood forming organs, Blood, Skeletal muscle, Lymphoid tissue, Spleen
Repeated Dose Toxicity Comments: PF-07302048: * Doses were administered once a week.
Reproduction & Development Toxicity: (Duration, Species, Route, Dose, End Point, Effect(s))
PF-07305885
Fertility & Embryonic Development - Females Rat Intramuscular 30 µg NOAEL No effects at maximum dose, Not
teratogenic
Potassium phosphate
Reproductive & Fertility Rat No route specified 282 mg/kg/day NOAEL No evidence of impaired fertility or harm to the fetus
Reproductive & Fertility Mouse No route specified 320 mg/kg/day NOAEL No evidence of impaired fertility or harm to the
fetus
Genetic Toxicity: (Study Type, Cell Type/Organism, Result)
Potassium phosphate
Bacterial Mutagenicity (Ames) Salmonella Negative
Carcinogenicity See below
Cholesterol
IARC Group 3 (Not Classifiable)
Data for the Drug Product
_____________________________________________________________________________________________
PF00092

SAFETY DATA SHEET
Product Name Pfizer-BioNTech COVID-19 Vaccine Page 9 / 12
Revision date 07-Dec-2021 Version 3
_____________________________________________________________________________________________
Reproduction & Development Toxicity: (Study Type, Species, Route, Dose, End Point, Effect(s))
Fertility & Embryonic Rat Intramuscular N/A Not specified No effects at
Development - Females maximum dose
11.2. Information on other hazards
11.2.1. Endocrine disrupting properties
Endocrine disrupting properties No information available.
11.2.2. Other information
Other adverse effects No information available.
Section 12: ECOLOGICAL INFORMATION
Environmental Overview: Environmental properties have not been investigated. Releases to the environment should
be avoided.
12.1. Toxicity
Aquatic Toxicity: (Species, Method, End Point, Duration, Result)
POTASSIUM CHLORIDE
Gambusia affinis (Mosquitofish) LC50 96 hours 920 mg/L
Lepomis macrochirus (Bluegill Sunfish) LC50 96 hours 2010 mg/L
Daphnia Magna (Water Flea) EC50 48 hours 825 mg/L
Scenedesmus subspicatus (Green Alga) EC50 72 hours 2500 mg/L
12.2. Persistence and degradability
Persistence and degradability No information available.
12.3. Bioaccumulative potential
Bioaccumulation No information available.
12.4. Mobility in soil
Mobility in soil No information available.
12.5. Results of PBT and vPvB assessment
PBT and vPvB assessment .
Chemical name PBT and vPvB assessment
SODIUM CHLORIDE The substance is not PBT / vPvB PBT assessment does
not apply
Potassium phosphate The substance is not PBT / vPvB PBT assessment does
not apply
POTASSIUM CHLORIDE The substance is not PBT / vPvB PBT assessment does
not apply
Cholesterol The substance is not PBT / vPvB
12.6. Endocrine disrupting properties
_____________________________________________________________________________________________
PF00092

SAFETY DATA SHEET Product Name Pfizer-BioNTech COVID-19 Vaccine Page 10 / 12 Revision date 07-Dec-2021 Version 3 _____________________________________________________________________________________________ Endocrine disrupting properties No information available. 12.7. Other adverse effects No information available. Section 13: DISPOSAL CONSIDERATIONS 13.1. Waste treatment methods Dispose of waste in accordance with all applicable laws and regulations. Member State specific and Community specific provisions must be considered. Considering the relevant known environmental and human health hazards of the material, review and implement appropriate technical and procedural wastewater and waste disposal measures to prevent occupational exposure and environmental release. It is recommended that waste minimization be practiced. The best available technology should be utilized to prevent environmental releases. This may include destructive techniques for waste and wastewater. Section 14: TRANSPORT INFORMATION The following refers to all modes of transportation unless specified below. Not regulated for transport under USDOT, EUADR, IATA, or IMDG regulations. Section 15: REGULATORY INFORMATION 15.1. Safety, health and environmental regulations/legislation specific for the substance or mixture Water CERCLA/SARA Section 313 de minimus % Not Listed California Proposition 65 Not Listed TSCA Present EINECS 231-791-2 AICS Present Sucrose CERCLA/SARA Section 313 de minimus % Not Listed California Proposition 65 Not Listed TSCA Present EINECS 200-334-9 AICS Present SODIUM CHLORIDE CERCLA/SARA Section 313 de minimus % Not Listed California Proposition 65 Not Listed TSCA Present EINECS 231-598-3 AICS Present ALC-0315 CERCLA/SARA Section 313 de minimus % Not Listed California Proposition 65 Not Listed EINECS Not Listed Potassium phosphate CERCLA/SARA Section 313 de minimus % Not Listed California Proposition 65 Not Listed TSCA Present EINECS 231-913-4 _____________________________________________________________________________________________ PF00092










